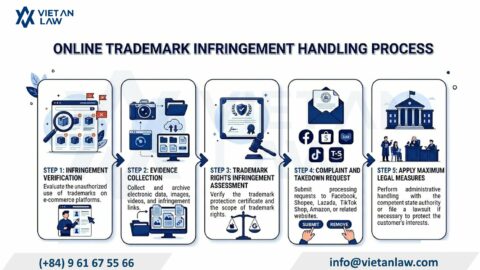
Adapting to Vietnam’s rapidly evolving regulatory landscape is essential for businesses maintaining market compliance. The Law on Quality of Products and Goods 2007 has long served as the fundamental legal framework governing product standards in Vietnam. However, following the National Assembly’s approval of the amended Law on June 18, 2025, the Government issued Decree 37/2026/ND-CP to provide detailed guidance and align with practical international demands. Effective from January 23, 2026, this Decree replaces Decree 132/2008/ND-CP. Below, Viet An Law provides an update on the New Highlights of Decree 37/2026/ND-CP on Product and Goods Quality.
Decree 37/2026/ND-CP specifies the principles for identification, criteria for risk assessment, methods for determining risk levels, and the responsibilities for evaluating and updating the list of medium and high-risk products. This risk classification approach enhances transparency and consistency while reducing the administrative burden on products with low risk levels.
Under Clause 2, Article 6 of the Decree, products and goods are categorized into three distinct levels:
Chapter II of Decree 37/2026/ND-CP provides specific regulations on investment and support for testing and certification organizations to achieve international standards. It encourages the mobilization of social resources and enterprise participation in building the National Quality Infrastructure (NQI).
The State will focus investment on infrastructure, technology, and key industrial sectors, while strengthening international cooperation mechanisms to attract investment capital and R&D projects. As the foundation for ensuring the quality of products, goods, and services, the NQI is vital for enhancing national competitiveness. The centralized management of the NQI prevents overlap and optimizes resources in accordance with international practices from ISO, OIML, and ILAC.
For the first time, Article 16 of Decree 37/2026/ND-CP introduces a detailed concept and regulations for quality controllers. Accordingly, a quality controller is defined as a “civil servant, public employee, or officer in the people’s armed forces who is assigned, arranged, or transferred to a job position and ranked as a ‘quality controller’ within a product and goods quality inspection agency in accordance with the law.”
This new regulation expands the scope of controllers beyond traditional civil servants to include public employees and military officers, matching the broad operational scope of quality inspection activities. It also clarifies operational principles and responsibilities to ensure transparency and professionalism, while adding provisions for uniforms, identification cards, and professional standards.
The Decree introduces the concept of electronic labeling as a modern alternative to traditional physical labels. Under Clause 4, Article 3, an electronic label is “a goods label in electronic form expressed through a data carrier that is pasted, printed, attached, molded, engraved, mounted, or displayed directly on the goods or the commercial packaging of the goods.”
Article 32 of Decree 37/2026/ND-CP strongly encourages the transition from physical labels to electronic labeling and digital passports. Enterprises now have the right to use electronic labels to display all mandatory content, except in cases where specialized laws specifically require physical labeling. The digital product passport acts as a digital identity card, storing information on origin and quality standards, facilitating consumer lookup and assisting e-commerce platforms in maintaining transparency.
Article 18 of Decree 37/2026/ND-CP regulates the application of barcodes in the quality management and traceability of products and goods.
The Decree encourages manufacturing and trading organizations to apply barcodes and utilize technological platforms such as Blockchain, Internet of Things (IoT), and Artificial Intelligence (AI) to meet their specific management needs. Once an organization chooses to apply barcodes for traceability and quality management, it must comply with the relevant procedural requirements set forth in this Decree.
Decree 37/2026/ND-CP brings practical changes to State inspection activities:
Under the new regulations, traceability activities must adhere to four core principles: data sharing, availability of master data elements, transparency, and full participation of all parties.
Traceability data for products categorized as high-risk must be connected and shared with the National Product and Goods Traceability Portal. This synchronization between State management agencies and enterprises is a critical step in handling quality incidents rapidly and ensuring a unified data ecosystem.
Article 12 of Decree 37/2026/ND-CP stipulates that the State shall allocate an annual budget to develop the NQI and provide direct support to businesses:
In summary, the New Highlights of Decree 37/2026/ND-CP on Product and Goods Quality represent a significant shift toward digitalization and risk-based management. These changes aim to modernize Vietnam’s quality control systems while providing substantial support for enterprises to enhance their global competitiveness.
For detailed advice or assistance regarding compliance with the new Decree, please contact Viet An Law for professional support.